👀 DON'T MISS 👀
"I almost grew a fear of flying," said the former quality inspector for one of Boeing's main suppliers, Spirit AeroSystems.
The president urged the airplane manufacturer to reach a deal that "gets these firefighters the pay and benefits they deserve."
The "Morning Joe" co-host mocked Trump's dinner with buyers of his digital trading cards on a day off from court.
Los Angeles Superior Court Judge Gus T. May said the rock legend needs the conservatorship because of what his doctor calls a “major neurocognitive disorder.”
Scott's absence in the Senate did not affect the legislative outcome, but it's the latest sign of Republican devotion to the former president.
Environmental groups have petitioned the White House to establish a national monument along the Dolores River — and many local residents are fuming.
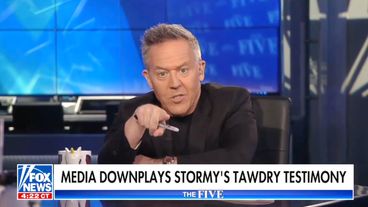
The Fox News host offered a deeply misogynistic take on the adult film star's testimony about her alleged affair with Trump.
The EGOT winner recently fulfilled her decadelong quest to meet one world-renowned figure — and even handed him a copy of the "Sister Act" soundtrack.
The former Hawaii congresswoman said she hadn't, but refused to throw Kristi Noem under the bus.
The senator, who became a meme over her SOTU rebuttal earlier this year, cast Donald Trump as a role model for kids.
Seinfeld made the remark during an appearance on the "Fly on the Wall" podcast.
Jack Schlossberg, Caroline Kennedy's kid, showed off his accents to bolster the family's opposition to the independent candidate.