By: Charles Q. Choi, InnovationNewsDaily Contributor
Published: 04/19/2012 04:26 PM EDT on InnovationNewsDaily
Synthetic molecules resembling DNA can function and evolve just like the real thing, its developers say.
These new, unnatural building blocks could be more useful than DNA or its closely related biomolecule, RNA, in a variety of medical and biotechnology applications, researchers added. Other investigators noted they could even lead to novel forms of life.
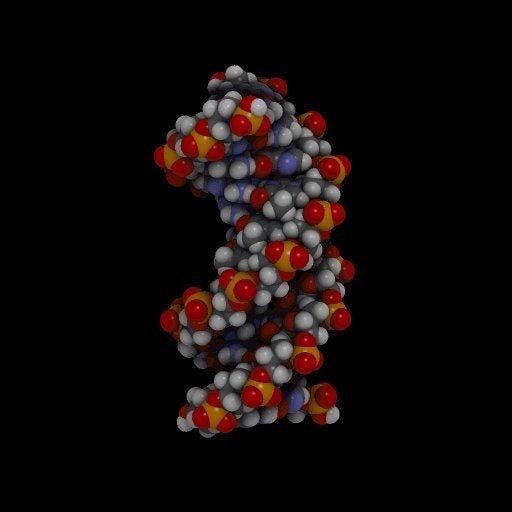
DNA is essentially made of four different kinds of molecules known as nucleic acids, commonly referred to by their initials, A, G, C and T. These run along a backbone made of sugars and phosphate groups.
Scientists call their artificial nucleic-acidlike molecules XNA, in which the natural sugar component has been replaced by one of six alternative organic compounds. These XNA molecules all can bind to DNA and RNA.
The researchers also have developed enzymes that can synthesize XNA from a DNA template, plus others that can "reverse transcribe" XNA back into DNA. This means they can store and copy data just as DNA can — the basis of heredity for all life on Earth.
The investigators subjected an XNA molecule to artificial natural selection in the lab by introducing mutations into its genetic code. By allowing the different versions of the molecule to compete against each other for binding to another molecule, the team ended up with a shape that bound tightly and specifically to the target – just as one would expect of DNA under the same conditions. This makes XNA the only known molecules other than DNA and RNA capable of Darwinian evolution.
"Heredity — information storage and propagation — and evolution, two of the hallmarks of life, can be implemented in polymers other than DNA and RNA," researcher Philipp Holliger at the Medical Research Council Laboratory of Molecular Biology in Cambridge, England, told InnovationNewsDaily.
One notable property of XNA molecules is they are not biodegradable: They are impervious to natural enzymes that degrade DNA and RNA. As such, they could find use in medical and biotechnology arenas where DNA and RNA could not go.
"People use RNA and DNA for biotechnology, therapeutics, diagnostics and biosensing applications, but these are very fragile in the face of biology — they degrade very rapidly if there's any contact with biological materials," said biochemist Gerald Joyce at the Scripps Research Institute, who did not take part in this work. "As such, chemists have to work to make them resistant to natural enzymes that degrade RNA and DNA, and then you have to worry about losing the good properties of those molecules. These XNAs, however, are resistant from the get-go."
These findings might also shed light on the origins of life — specifically, why DNA and RNA came to dominate Earth.
"It shows that there is no overwhelming functional imperative for life to use DNA and RNA for genetic information storage and propagation. More likely, this choice reflects a 'frozen accident' from the origin of life," Holliger suggested.
The construction of genetic systems based on alternative chemical platforms may ultimately lead to the synthesis of novel forms of life, if researchers can devise a system for XNA to replicate itself just as DNA has, Joyce said. However, he cautioned that synthetic biologists should take care to "not tread into areas that have the potential to harm our biology." For instance, the fact that XNA is not biodegradable suggests that life might not have any easy way of breaking it down.
"Do I think what these researchers have done is dangerous? Absolutely not. Do I think this is going to be dangerous in the near or even medium term? Absolutely not," Joyce said. "Still, are we treading into something risky here? It's synthetic biology, not a natural form of biology. Scientists have to pay attention here."
The scientists detailed their findings in the April 20 issue of the journal Science.
Copyright 2012 InnovationNewsDaily, a TechMediaNetwork company. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.
Our 2024 Coverage Needs You
It's Another Trump-Biden Showdown — And We Need Your Help
The Future Of Democracy Is At Stake
Our 2024 Coverage Needs You
Your Loyalty Means The World To Us
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
The 2024 election is heating up, and women's rights, health care, voting rights, and the very future of democracy are all at stake. Donald Trump will face Joe Biden in the most consequential vote of our time. And HuffPost will be there, covering every twist and turn. America's future hangs in the balance. Would you consider contributing to support our journalism and keep it free for all during this critical season?
HuffPost believes news should be accessible to everyone, regardless of their ability to pay for it. We rely on readers like you to help fund our work. Any contribution you can make — even as little as $2 — goes directly toward supporting the impactful journalism that we will continue to produce this year. Thank you for being part of our story.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
It's official: Donald Trump will face Joe Biden this fall in the presidential election. As we face the most consequential presidential election of our time, HuffPost is committed to bringing you up-to-date, accurate news about the 2024 race. While other outlets have retreated behind paywalls, you can trust our news will stay free.
But we can't do it without your help. Reader funding is one of the key ways we support our newsroom. Would you consider making a donation to help fund our news during this critical time? Your contributions are vital to supporting a free press.
Contribute as little as $2 to keep our journalism free and accessible to all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. Would you consider becoming a regular HuffPost contributor?
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. If circumstances have changed since you last contributed, we hope you'll consider contributing to HuffPost once more.
Already contributed? Log in to hide these messages.