This story originally appeared at Fast Company's Co.Design.
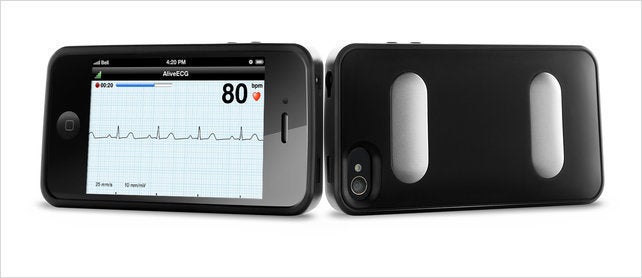
Most iPhone cases just protect your phone from drops. If you’re getting fancy, it may have a fisheye camera lens or a screen-printed back. But what about diagnosing coronary heart disease, arrhythmia, or congenital heart defects? The AliveCor Heart Monitor is an FDA-approved iPhone case that can be held in your hands (or dramatically pressed against your chest) to produce an EKG/ECG--the infamous green blips pulsing patient-side in hospitals everywhere.
“We think that EKG screening can be as approachable as taking blood pressure,” AliveCor President and CEO Judy Wade tells Co.Design.
There are already apps that take your heartbeat, of course. But there’s a big difference between the fast-paced standards of casual electronics and the strict sanctions of government-approved medical devices. “The heartbeat camera apps are good at wellness,” Wade admits, “but we see ourselves for use by people who want clinical-quality equipment.”
So unlike most iPhone cases that are squirted by Chinese factories at extremely high margins, AliveCor’s case has been in serious development since 2010. Aside from building the gadget itself, to become approved for medical use by the FDA, AliveCor had to participate in two clinical trials to field test both the hardware and the accompanying app. One study investigated how its single-lead EKG compared to a traditional 12-lead device, the other examined if 54 participants could figure out how to use the case properly, with no previous medical training. The latter study was not only successful but led to the diagnosis of two serious heart problems.
The Complications Of Innovating Under The FDA
AliveCor was lucky. Though it took about six months to get the application ready, the approval arrived well within the 90-day approval window, allowing the company to come to market sooner. It was a necessary hassle; FDA approval opens a lot of doors. Instantly, what could be considered some scam iPhone case was marketable to health care professionals--doctors--who’d most likely pay out of pocket for a $200 stethoscope replacement without blinking. FDA approval also allows doctors to prescribe, and potentially have insurance cover, AliveCor’s device for their patients to take home.
More from Co.Design:
Would Netflix Live Happily Ever After Without Disney Content?
Naked Bodies In Motion: Safe For Work, Artfully Abstract
But even with an approval in-hand, AliveCor will continue to juggle complicated regulations to stay competitive in the market. For one, the approved monitor was designed for the iPhone 4 and 4S. Before AliveCor can release an iPhone 5 version with the exact same hardware internals, they will need to seek out additional FDA approval. (With previous approval and clinical trials to cite, the process is mostly a formality, but it’s still paperwork that takes more time and resources.)
The company also intends to release an over-the-counter version of the case. The good news is, this device will be eligible for coverage in most employee spending programs. But because of FDA regulations, this OTC version cannot provide the raw EKG data to a consumer who might not know how to interpret the esoteric waveforms. Instead, AliveCor will redesign the app to provide an infographic-esque interpretation of the EKG. “An EKG means something to a trained physician, but we can provide a lot of insights to an untrained consumer that might help explain what triggered a cardiac event,” Wade explains. “Like caffeine is a trigger. With an app, we see being able to offer more insight to an individual about their heart health.”
From a product design standpoint, this second-level data analysis sounds like an ideal, consumer-oriented decision. But from a consumer rights standpoint, why is any government agency standing in the way of consumer access to our own raw data? I can see how strongly my iPhone’s antenna is reaching the nearest cell tower, but I can’t see how well my own heart is ticking inside my body? How absurd is that? Interestingly enough, AliveCor is using this regulation to their advantage, banking on the health care model as it stands now. Its OTC device will offer services to refer you to a physician for deeper result analysis (and access to your actual waveforms, if you’re so concerned), which will provide a backend revenue stream beyond typical hardware sales. Imagine the potential: In-app purchase for a follow-up appointment.
The Future Of Medicine
For the time being, AliveCor is continuing to develop their EKG cases into a full line, including that OTC device, which will also be a universal version working for both iOS and Android. (Since the case actually communicates with the phone wirelessly, once the software programming is done, these product differentiations are largely cosmetic in nature.) No doubt, AliveCor sees the case as a stepping stone to the company’s overall vision, that “everyone should have their health at their fingertips,” Wade says. But the company will have to solve a lot of larger problems that the industry is struggling with to make that future a reality.
While diagnostic devices may be coming to the phone, we still have no standards to get such diagnostic information back to our doctors. AliveCor explained to me that it can send a push notification to my cardiologist every time I check my heart, but does my cardiologist really want push notifications all day from their client list? Or worse, would any doctor want a devastating cardiac episode just sitting under 30 other messages in the iOS Notification Center? Should my phone text or not text emergency information? Should doctors be held accountable for app-based information? Should medical devices be regulated to automatically dial 911 in cases of emergency?
No doubt, AliveCor’s Heart Monitor is another case of affordable consumer technology outpacing our brick-and-mortar hospitals, but to the credit of our hospitals, affordable consumer technology is outpacing most of the world. Still, just as Domino’s has figured out to deliver me a pizza through an app (no doubt, saving a few cents in the process), so, too, will the medical community come around to juggling big data at the individual patient level. The real question is, will FDA regulations leave space for the little guys--the weekend app warriors and the Kickstarters--to innovate responsibly, at a price cheaper than clinical trials and a timeframe faster than paperwork?
Read this story and more at Fast Company's Co.Design.