Suppose you are sending your 12-year-old daughter off to a summer camp two states away and she is flying on a plane by herself for the first time. Would you put her on that plane if you knew that it had less than a 10 percent chance of landing safely at its destination?
Why do we accept these odds when it comes to animal experimentation? If you were volunteering for a clinical trial, there is more than 90 percent chance that the drug that tested safe and effective in animals will be ineffective or unsafe in you.
In the previous blogs in my series on animal experimentation, we reviewed the three main reasons why animal experimentation doesn't work. Unlike failures in many other scientific areas, the failure of animal experimentation has a high cost -- and harms us in three crucial ways:
1. Misleading Safety Tests in Animals Directly Hurt Humans.
In March, 2006, six human volunteers were injected with TGN 1412, an experimental therapy created by TeGenero. As described by Slate:
Within minutes, the human test subjects were writhing on the floor in agony. The compound was designed to dampen the immune response, but it had supercharged theirs, unleashing a cascade of chemicals that sent all six to the hospital. Several of the men suffered permanent organ damage, and one man's head swelled up so horribly that British tabloids refer to the case as the 'elephant man trial'.
TGN 1412 was tested in mice, rabbits, rats and monkeys with no ill effects. In addition, cynomolgus monkeys were used because they best replicated the human mechanisms specifically targeted by TGN 1412 (1). Thus, not only were several different species used, those deemed most relevant to humans were used. Monkeys also underwent repeat-dose toxicity studies and in fact were given 500× the dose given to the human volunteers for not less than four consecutive weeks. Still, none of the monkeys manifested the ill effects that humans showed within minutes of receiving a miniscule amount of the test drug.
TGN 1412 exemplifies how animal experiments are notably unreliable for determining if a chemical or drug will be safe in humans. Here are some other examples where animal experiments hurt people:
- In 2003, Élan Pharmaceuticals had to stop trials of an Alzheimer's vaccine that had cured the disease in "Alzheimer's mice," after the substance caused brain inflammation in humans (2).
A highly touted gene therapy that cured dogs of hemophilia was discontinued because it caused liver damage and other problems in humans that were not seen in animal experiments. An NIH clinical trial of fialuridine, a promising medication for Hepatitis B, was abruptly terminated because it caused liver failure in seven out of ten people, five of whom died and two of whom required liver transplants (3). Thus, far from protecting us, animal experimentation puts us at greater risk of harm. Additionally, the indirect harms as a result of misleading tests can be substantial.
2. Misleading Animal Experiments May Cause Us to Throw Away Cures
It is hard to quantify how many missed opportunities there may have been because of misleading animal experiments. However, there are plenty of examples that demonstrate how lucky we are that researchers did not believe the animal tests. For example:
- An editorial in Nature Reviews Drug Discovery describes how tamoxifen, one of our most effective drugs against certain types of breast cancer, would have been abandoned because it causes liver tumors in rats, a problem that does not carry over to humans.
As Dr. John Pippin argues:
Gleevec is a success of rational drug design and human-based drug testing -- a life-prolonging success that would have been lost if the results of animal research had prevailed.
- Experiments on animals delayed the acceptance of cyclosporine, a drug widely and successfully used to treat autoimmune disorders and prevent organ transplant rejection (5).
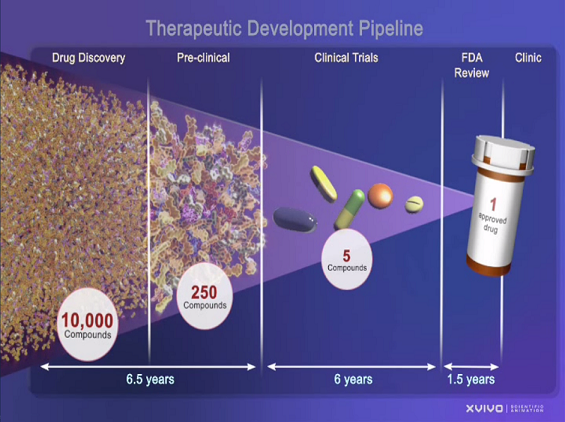
Of every five to 10,000 potential drugs tested in the lab, only about five pass on to clinical trials. Many don't pass the animal tests because of species-specific results. Yet many of these agents would likely have worked spectacularly and been safe in humans.
One can't help but wonder how many people would have been saved if human-based tests that would better predict harmful effects and drug efficacy were used rather than animal experiments?
And so we come to the last major harm.
3. Time and Money Wasted on Animal Experiments Could Have Been Directed into More Fruitful Human-Based Tests.
An invalid disease model can lead the industry in the wrong direction, wasting time and significant investment. On average, it costs a company more than $1 billion to get one new drug to the market. NIH alone spends almost half of its funding -- up to 14.5 billion of our tax dollars yearly -- on animal experiments.
Repeatedly, researchers have been lured down the wrong line of investigation because of information gleaned from animal experiments that later proved to be inaccurate, irrelevant or contrary to human biology. It's taken over 25 years of failed HIV vaccine clinical trials for researchers to seriously question the usefulness of non-human primate HIV experiments, and over 30 years before we realized that the rodent model of diabetes is wrong.
We can be pretty confident that the victims of the TGN 1412 disaster will never risk their lives based on animal experiments again. In fact, a human-based in vitro test would have predicted the harmful effects and protected those men (7).
How many more people need to suffer and die before we realize that, if we really want to help ourselves, we need to cut out the animal experiments and focus on more effective human-based tests?
Stay tuned for the next blog in the series!
Want to know more? Check out my website and join me on Facebook.
- Akhtar (2012) Animals and public health. Why treating animals better is critical to human welfare. Hampshire, UK: Palgrave Macmillan, p. 147-148.
Allen. Of mice and men. The problems with animal testing. Slate. June 1, 2006. Mckenzie, et al. Hepatic Failure and Lactic Acidosis Due to Fialuridine (FIAU), an Investigational Nucleoside Analogue for Chronic Hepatitis B. N Engl J Med 1995; 333:1099-1105. Pippin. South Texas Law Review 2013; 54: 469-511. Greek, Greek. Animal research and human disease. JAMA 2000; 283: 743-744. Ostrovsky, et al. Vision following extended congenital blindness.Psychological Science 2006; 17: 1009-1014 Dhir et al. A predictive biomimetic model of cytokine release induced by TGN1412 and other therapeutic monoclonal antibodies. J. Immunotoxicol. 2012;9:34-42.Our 2024 Coverage Needs You
It's Another Trump-Biden Showdown — And We Need Your Help
The Future Of Democracy Is At Stake
Our 2024 Coverage Needs You
Your Loyalty Means The World To Us
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
The 2024 election is heating up, and women's rights, health care, voting rights, and the very future of democracy are all at stake. Donald Trump will face Joe Biden in the most consequential vote of our time. And HuffPost will be there, covering every twist and turn. America's future hangs in the balance. Would you consider contributing to support our journalism and keep it free for all during this critical season?
HuffPost believes news should be accessible to everyone, regardless of their ability to pay for it. We rely on readers like you to help fund our work. Any contribution you can make — even as little as $2 — goes directly toward supporting the impactful journalism that we will continue to produce this year. Thank you for being part of our story.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
It's official: Donald Trump will face Joe Biden this fall in the presidential election. As we face the most consequential presidential election of our time, HuffPost is committed to bringing you up-to-date, accurate news about the 2024 race. While other outlets have retreated behind paywalls, you can trust our news will stay free.
But we can't do it without your help. Reader funding is one of the key ways we support our newsroom. Would you consider making a donation to help fund our news during this critical time? Your contributions are vital to supporting a free press.
Contribute as little as $2 to keep our journalism free and accessible to all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. Would you consider becoming a regular HuffPost contributor?
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. If circumstances have changed since you last contributed, we hope you'll consider contributing to HuffPost once more.
Already contributed? Log in to hide these messages.