Food and Drug Administration
About one in five samples of commercially sold pasteurized milk contained traces of the highly pathogenic avian influenza.
"I’m 70 years old and we were talking about this when I was in college, and that's insane to me," said Jane Amgarola, a retiree who speaks for countless others.
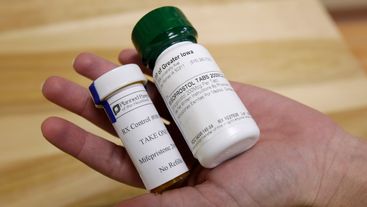
A group of more than 260 members of Congress also submitted a friend-of-the-court brief ahead of the mifepristone hearing in March.
A drug called tianeptine, or "gas station heroin," has been found in over-the-counter “Neptune's Fix” supplements.
WHAT'S HAPPENING
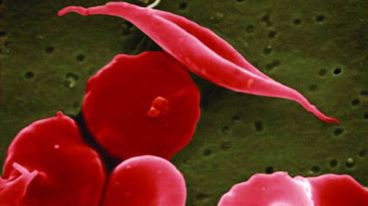
The painful, inherited blood disorder afflicts mostly Black people in the U.S.
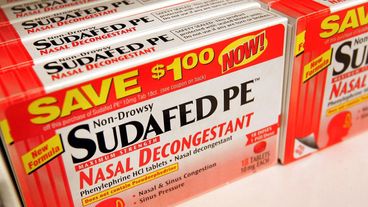
Phenylephrine, which is found in some Sudafed, Mucinex, Benadryl and Vicks products, is no better than a placebo, said an independent advisory committee to the FDA.
Taking the contraceptives "could result in unexpected pregnancy," the Food and Drug Administration said.
Last year, a surge in RSV cases flooded U.S. hospitals with wheezing children.
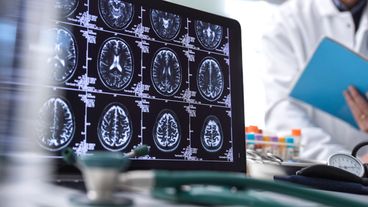
Experts say the medicine is an exciting development for treating memory loss — but it does come with risks.
The FDA is currently deciding whether to make one brand of oral contraceptives available at pharmacies without a prescription.