By: Rachael Rettner
Published: 11/03/2014 10:12 AM EST on LiveScience
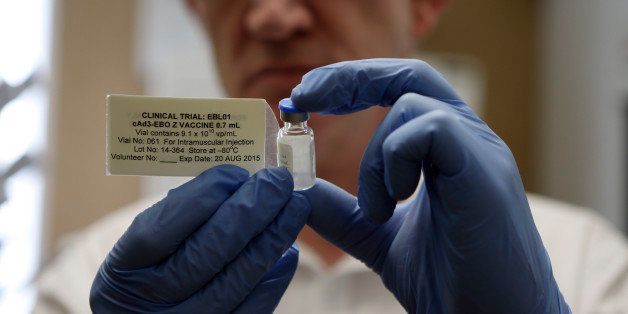
Two experimental vaccines against Ebola are currently being tested to see whether they are safe to use in people, and health officials have said that millions of doses could be available by the end of next year. But how do the vaccines work?
Both vaccines essentially consist of a harmless virus that has been "spiked" with a protein from the Ebola virus, said Derek Gatherer, a bioinformatics researcher at Lancaster University in the United Kingdom who studies viral genetics and evolution.
If a person is given the vaccine, "the body thinks it's being infected with this rather innocuous virus, [and] part of the virus happens to be the Ebola protein," said Gatherer, who is not involved in work on the Ebola vaccines. This prompts an immune response, and the body develops antibodies against the Ebola protein, Gatherer said. [How Do People Survive Ebola?]
Ideally, if a vaccinated person were later exposed to the real Ebola virus, these antibodies would be ready to fight off the infection before it could take hold.
The first vaccine, which began safety testing this summer, is being developed by the National Institute of Allergy and Infectious Diseases (NIAID) and GlaxoSmithKline. It consists of a type of cold virus called an adenovirus that affects chimpanzees and has genetic material from two strains of Ebola: Zaire Ebola (which is causing the current outbreak in West Africa) and Sudan Ebola, according to NIAID.
The engineered adenovirus can't replicate in the human body. It's used to deliver the Ebola gene to a person's cells, which, in turn, produce a single Ebola protein. If the vaccine works as it should, this protein will cause an immune response. But in any case, it cannot cause Ebola virus disease, according to the NIAID.
The first test of the vaccine in people, called a Phase 1 trial, includes 20 healthy adults who will be injected with one of two doses of the vaccine.
The second vaccine, called VSV-ZEBOV, consists of a virus that mainly infects animals (including rodents, cattle, swine and horses), called the vesicular stomatitis virus (VSV). In the vaccine, one gene of VSV has been replaced with the gene for the outer protein of the Zaire Ebola virus, according to the National Institutes of Health.
Safety testing of the VSV-ZEBOV vaccine began this month at the NIH, the agency said. The study involves 39 healthy adults who will be given either a low dose of the vaccine, a higher dose of the vaccine or a placebo. VSV-ZEBOV was developed by the Public Health Agency of Canada and was licensed to the biopharmaceutical company NewLink Genetics Corp.
Results of the safety trials are expected by December, and the second round of trials will begin in people in West Africa shortly thereafter, according to the World Health Organization. (The second round of testing is known as Phase 2 trials, which will further test vaccines' safety, and also look at its effectiveness.)
If the vaccines are effective, pharmaceutical companies could manufacture several hundred thousand doses in the first half of 2015, and millions of doses by the end of that year, WHO said.
Follow Rachael Rettner @RachaelRettner. Follow Live Science @livescience, Facebook & Google+. Original article on Live Science
Copyright 2014 LiveScience, a TechMediaNetwork company. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.
Our 2024 Coverage Needs You
It's Another Trump-Biden Showdown — And We Need Your Help
The Future Of Democracy Is At Stake
Our 2024 Coverage Needs You
Your Loyalty Means The World To Us
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
The 2024 election is heating up, and women's rights, health care, voting rights, and the very future of democracy are all at stake. Donald Trump will face Joe Biden in the most consequential vote of our time. And HuffPost will be there, covering every twist and turn. America's future hangs in the balance. Would you consider contributing to support our journalism and keep it free for all during this critical season?
HuffPost believes news should be accessible to everyone, regardless of their ability to pay for it. We rely on readers like you to help fund our work. Any contribution you can make — even as little as $2 — goes directly toward supporting the impactful journalism that we will continue to produce this year. Thank you for being part of our story.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
It's official: Donald Trump will face Joe Biden this fall in the presidential election. As we face the most consequential presidential election of our time, HuffPost is committed to bringing you up-to-date, accurate news about the 2024 race. While other outlets have retreated behind paywalls, you can trust our news will stay free.
But we can't do it without your help. Reader funding is one of the key ways we support our newsroom. Would you consider making a donation to help fund our news during this critical time? Your contributions are vital to supporting a free press.
Contribute as little as $2 to keep our journalism free and accessible to all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
As Americans head to the polls in 2024, the very future of our country is at stake. At HuffPost, we believe that a free press is critical to creating well-informed voters. That's why our journalism is free for everyone, even though other newsrooms retreat behind expensive paywalls.
Our journalists will continue to cover the twists and turns during this historic presidential election. With your help, we'll bring you hard-hitting investigations, well-researched analysis and timely takes you can't find elsewhere. Reporting in this current political climate is a responsibility we do not take lightly, and we thank you for your support.
Contribute as little as $2 to keep our news free for all.
Can't afford to donate? Support HuffPost by creating a free account and log in while you read.
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. Would you consider becoming a regular HuffPost contributor?
Dear HuffPost Reader
Thank you for your past contribution to HuffPost. We are sincerely grateful for readers like you who help us ensure that we can keep our journalism free for everyone.
The stakes are high this year, and our 2024 coverage could use continued support. If circumstances have changed since you last contributed, we hope you'll consider contributing to HuffPost once more.
Already contributed? Log in to hide these messages.