Story comes courtesy of California Watch.
Southern California’s largest clinical laboratory and radiology company serving nursing homes faces fraud allegations, adding to the growing number of False Claims Act lawsuits filed against the health care industry nationwide.
According to court documents, Burbank-based Diagnostic Labs, which offers medical lab tests and radiology services, provided deep discounts to skilled nursing facilities in exchange for business that can be charged to taxpayer-funded Medicare and Medi-Cal.
“This is an illegal kickback scheme, no more legal than if Diagnostic Labs, rather than providing below-cost discounts, had instead simply handed the SNFs (skilled nursing facilities) an envelope of cash,” the lawsuit alleges.
Attorneys for Diagnostic Labs did not respond to requests for comment by phone and email. In court documents, they described the allegations as overblown.
The case against Diagnostic Labs was brought under the False Claims Act and a parallel state statute, both of which make it illegal to defraud the government. These laws are increasingly invoked in the health care arena, legal observers said.
“Since the start of the Obama administration, there has been a pretty pronounced and noticeable uptick in government investigations in which the False Claims Act is the spearhead of the investigation,” said Zack Buck, a visiting assistant professor at Seton Hall Law School who teaches a class on health care fraud and abuse. “The government has put a lot of money into fraud investigations and prosecutions and as a result, the amount of settlements have grown and continues to increase.”
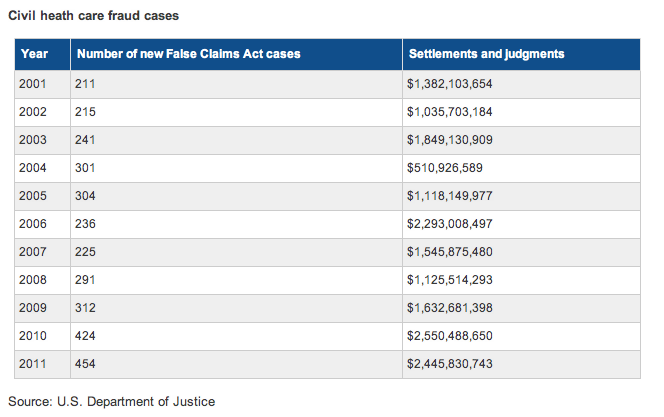
According to a statement made recently by Stuart F. Delery, acting assistant attorney general of the civil division of the U.S. Department of Justice, more than 630 whistleblower lawsuits were filed with the Department of Justice in 2011 – more than any other year and an increase of nearly 50 percent since 2009. More than two-thirds of these cases are related to allegations of fraud related to government health care programs.
These lawsuits have produced significant financial results for the federal government.
In 2011, the federal government collected $2.4 billion in settlements and judgments from health care cases – more than from any other industry, according to an analysis by law firm Gibson, Dunn & Crutcher. Over the past three years, the government has collected $6.6 billion in relation to health care fraud.
A majority of these funds are returned to the relevant federal or state-funded health programs.
Given the significant financial recoveries connected to health care-related false claims cases, the industry will continue to receive regulatory scrutiny, Buck said.
“There’s been an explosion in attention and realization that we’ve got to do something with our health care program if it is going to survive – things need to be trimmed and cut and going after fraud is a good way to do it,” Buck said. “It’s a good investment.”
The Diagnostic Labs lawsuit was brought by two former employees. They claim that Diagnostic Labs exploited the Medicare and Medi-Cal billing system by charging nursing homes as little as $1 per patient per day for lab and radiology services. These facilities then referred additional outpatient lab tests or X-ray work to Diagnostic, which billed the federal and state governments the maximum amount government will pay for these services, court documents said.
The complaint alleged that as of March 2005, Diagnostic Labs offered a 71 percent discount to skilled nursing facilities for lab tests and a 69 percent discount for radiology services, and it “would have been impossible for Diagnostic Labs to stay in business” at the rates it charged the nursing homes if not for the “illegally induced” outpatient services that it billed to Medicare and Medi-Cal.
The case “signals a trend of greater scrutiny of the types of more subtle kickbacks that are common in the health care industry,” said Justin T. Berger, an attorney for the former employees. “There have always been kickbacks of all shapes and forms. What folks are more focused on now are these creative types of kickbacks.”
The case was filed in 2010 and unsealed in November 2011, and the state of California has joined the litigation.
In court documents, Diagnostic Labs said that the whistleblowers make “the leap that giving a ‘discount’ equals an intent to induce referrals.”
“There is no specific factual allegation tying any specific remuneration to any resulting referrals,” court documents filed by the company said.
Few False Claims Act cases go to trial, which means some areas remain legally murky, said Seton Hall’s Buck. “Diagnostic may have some right to offer discounts to their clients,” he said. “The question is, where do you cross the line and it becomes an inducement to refer business? That’s what’s sticky.”
Another False Claims Act case alleging similar practices was filed in Ohio involving a laboratory owned by Audax Group and Frazier Healthcare, the same venture capital firms that own Diagnostic Labs.
Diagnostic Labs does business with about 80 percent of the skilled nursing facilities in Southern California, said Berger, an attorney with Burlingame's Cotchett, Pitre & McCarthy.
Bernice Yeung is an investigative reporter for California Watch, a project of the non-profit Center for Investigative Reporting. Find more California Watch stories here.