By Dr. Emmanuel Mignot
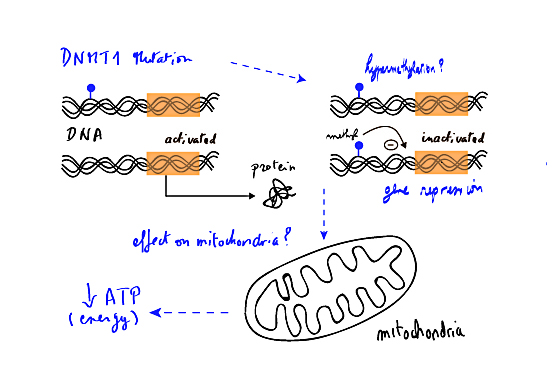
Illustration demonstrating some of the proposed mechanisms by which the gene mutation may lead to the ADCA-DN condition.
Previously, I mentioned how we managed to find the cause of Autosomal Dominant Cerebellar Ataxia, Deafness and Narcolepsy (or ADCA-DN) by sequencing all the genes of patients with the condition in three families. We discovered mutations in a particular gene called DNMT1 that controls other genes to silence their activity. Our discovery immediately led to an ethical conundrum, and spotlighted differences in medical and research practices between the U.S. and Europe.
First, we now had the ability to predict which family members will develop the condition in their lifetime. This was particularly difficult for us considering we had been in personal contact with family members from other countries. We had results showing, for example, that three young daughters in one family all were carrying the mutation.
In Italy and Sweden, doctors directly talked to the patients and evaluated who was ready and willing to get the results, but in the U.S., the process took over six months. Stanford's ethics and legal teams advised us to redirect the U.S. family members interested in knowing their genetic testing results to seek professional genetic counseling and to have an accredited facility in Chicago redo the testing, following Clinical Laboratory Improvement Amendments (CLIA). Interestingly, the American Medical Association (AMA) recently advised physicians in a statement to reveal to their patients any incidental finding found in the course of genetic research. All this had a cost of approximately $1,000 per person, and we decided to cover these expenses for the family, but the genetic counselors had long waiting lists and the Chicago lab had to repeat our tests using new blood samples. Eventually, however, the family was fully informed. Sadly, one of the main contacts in our U.S. family was also a mutation carrier, adding urgency to the situation.
Fortunately, however, we felt hopeful that our discovery had the possibility of leading to a treatment, most notably for the family members who are young and not yet affected with the problem. The disease has a late onset, suggesting there is only a minute dysregulation of the gene activity, and that maybe the process could be delayed or entirely prevented. Moreover, DNMT1 is an enzyme that has been well-studied by many researchers for countless years, as it is very ancient, being found even in plants. We know, for example, that the activity of the protein is dependent on a common food supplement called S-adenosyl-L-homocysteine (SAM), which works as a cofactor to promote its activity. Similarly, DNMT1 has been suggested to be very important in cancer. This is because the addition of a methyl group to substances (a process called methylation) can regulate the activity of genes, including cancer-causing oncogenes. In addition, DNMT1 inhibitors have been synthesized and are available, thus representing a potential treatment. The next step was to determine whether or not the mutation involved a decrease or increase in methylation, and to develop a process to test possible drugs.
All research takes time and money, and we unfortunately have neither for this project. Researchers from Stanford, Bologna, and Sweden all chipped in and figured out a way to collect skin biopsy samples on some of these patients and their relatives to develop skin (fibroblast) cell lines. We grew these cell lines and are now looking at methylation patterns and gene expression to determine whether an increase or decrease in methylation is at fault. We are also exploring which genes are affected downstream from the methylation. Our favorite hypothesis is that increased methylation at a specific DNA site creates the problem, and that a DNMT1 inhibitor could help. Dr. Plazzi, our Italian colleague, believes that the methylation defect alters the activity of mitochondria, explaining the similarities between ADCA-DN and other mitochondrial diseases. These state-of-the-art genetic studies are done in the laboratory of Dr. Alex Urban, whose office is next door to my laboratory.
After much effort, we had found an entity that was ready to fund some of this research, but because the company did not agree to cover administrative costs at Stanford while asking for intellectual property rights of any breakthrough generated, the grant was rejected. It is disheartening to have bureaucratic technicalities block progress, especially when all parties agree that it is unlikely any intellectual property would be generated by such research. We next plan to submit an application to the federal government to further this research. But it takes almost a year to get any funding if successful, and (because of the sequester) only 10 percent of submitted applications are ever funded. The probability of success is low. We are however still going forward, as this research may have implications not only for these patients but possibly for other conditions as well.
Although a solution is not yet found, this story illustrates the extraordinary potential that biological research has today to help individual patients. In 100 years, people will look at our time as the golden age of biology. In the next decade, many cancers will become chronic diseases, and nearly everybody's genome will be sequenced. As most diseases are caused by environmental factors acting on a susceptible genetic background, this knowledge will help prevent diseases in many cases. Yet this is occurring at a time when we are increasingly burdened by paperwork and regulations, a chronic lack of funding, and a decreasing pipeline of dedicated scientists.
One of the worst regulatory burdens for research is the Health Insurance Portability and Accountability Act of 1996 (HIPAA). This regulation started with good intentions -- to keep health information private -- and yet it costs billions. I have asked hundreds if they knew anyone who has been "personally harmed by the leakage of personal health information" and the only strongly positive answer was from a lawyer who said he knew of many cases... Although there is no denying that harm can occur -- for example, if health information is leaked to an employer -- in my opinion HIPAA is too conservative and has become a deterrent to conducting research. In California, the law is particularly stringent following an incident at UCLA Medical Center that compromised the privacy of former California First Lady Maria Shriver.
Adequately compensating the few people who are truly harmed would likely be more cost-effective. It is increasingly paperwork-intensive to get approval for research protocols as mundane as taking a blood sample or filling out a questionnaire. (Basic consent forms are typically 7-12 pages in length.) Every university wants to keep the privilege of approving the consent procedure, so that multiple layers of approval are needed, and different institutions do not always agree on the procedure to follow.
Despite the discouraging climate of funding and regulations, we will be doing our best to find a solution for these families, submitting grant applications and working hard to discover the cure that they deserve.
Dr. Mignot is the director of the Stanford Center for Sleep Sciences and Medicine. This Center is the birthplace of sleep medicine and includes research, clinical, and educational programs that have advanced the field and improved patient care for decades. To learn more, visit us at: http://sleep.stanford.edu/.
For more from the Stanford Center for Sleep Sciences and Medicine, click here.
For more on sleep, click here.